Issue's
Year 2019 | Month: June | Volume 6 | Issue 1
Natural Pigment Removal Capability of Carbon Derived from the ‘Golden Fibre’ 10.30954/2347-9655.01.2019.1A Comparative Study on the Effect of Vernonia amygdalina (Bitter Leaf) and Glibenclamide in the Treatment of Diabetes in Alloxan-Induced Diabetic Albino Wistar Rats
I. N. Ikhajiangbe Happy1, D. N. Ezejindu2, N. Ezeokafor Emmanuel1
1Department of Physiology, College of Health Sciences, Nnamdi Azikiwe University, Nnewi, Nigeria
2Department of Anatomy, College of Health Sciences, Nnamdi Azikiwe University, Nnewi, Nigeria
Submission: 20 July 2014; Revision: 10 Aug 2014; Acceptance: 10 Sep 2014
ABSTRACT
Aims:
The aim of this study is to compare the effect of bitter leaf (Vernonia Amygdalina) and glibenclamide in the treatment of diabetes in Alloxan-induced diabetic albino wistar rats.
Materials and Methods:
A total of 24 adult albino rats were used for the study. The rats were grouped into three groups of eight animals per group. Group A received 0.5 ml of distilled water and served as the control. Group B received 10 mg/kg/body weight of glibenclamide. Group C received 100 mg/kg/body weight of bitter leaf extract. Effect of bitter leaf and glibenclamide were observed at every 2 days for up to 2 weeks.
Results:
The result showed that the extract caused a decrease in blood glucose level on the fourth day but this was not statistically significant. Also, glibenclamide caused a decrease in the blood glucose level on the third day; this decrease was not statistically significant but slightly significant on the fourth day. On the sixth day, the extract caused a slightly significant decrease but when compared with the sixth day of glibenclamide, the decrease was very significant. From the eight day to the fourteenth day, the decrease in the blood glucose levels for group B and C became very significant. However, group C (glibenclamide) showed highly significant decrease when compared with group B (extract). Glibenclamide showed significant (p<0.05 or p<0.01) decrease in mean blood glucose level from day 8 to till end (day 14) as compared to bitter leaf extract.
Conclusion:
The work suggests that glibenclamide has a more potent hypoglycemic effect in alloxan-induced diabetic wistar rats when compared with the extract and may be more effective in the treatment of diabetics.
Key words: Diabetes, alloxan, glibenclamide, Wistar rats, diabetes, Blood glucose, Vernonia amygdalina
INTRODUCTION
Vernonia amygdalina commonly called bitter leaf is the most widely cultivated species of the genus Vernonia, which has about 1000 species of shrubs.[1] It belongs to the family Astaraceae. The leaves of V. amygdalina locally known as Nchanwu are widely used for food and medicinal purposes in South Eastern Nigeria. Their leaves are ethno medically believed to be effective for the treatment of diabetes, dysentery, gastrointestinal disorders and fever. They are also used for culinary purposes such that soups prepared with these leaves either in combination or individually, are believed to improve lactation.[2] It is vegetatively cultivated by stem cutting at an angle of 45°C and popular in most of West Africa countries including Nigeria, Cameroon, Gabon and Congo Democratic Republic. It was named after an English Botanist William Vernon. It is also referred to as ironweed. V. amygdalina is frequently found in gardens.[3] It can adapt to a variety of climates unlike other plants that are native to certain areas. It is grown in many countries, in savannah zones and cultivated fields.[4] Although most popularly used for food, it has also, been traditionally used for its medicinal properties.[5] True to its name, bitter leaf is bitter to taste, but surprisingly delicious in meals.[6]
Glibenclamide also known as glyburide is an antidiabetic drug in a class of medications known as sulfonylureas, closely related to sulfa drugs. It was developed in 1966 in a cooperative study between Boehringer Mannheim (now part of Roche) and Hoechst (now part of Sanofi-Aventis).[7]
MATERIALS AND METHODS
A total of 24 albino Wistar rats weighing between 150 g and 200 g were obtained in the pre-clinical Animal House of College of Health Sciences, Nnamdi Azikiwe University, Nnewi Campus. They were acclimatized for a period of 14 days and housed under standard laboratory conditions (29 ± 2°C temperature, 40-55% humidity, good ventilation) and had free access to water and diet (normal rat chow) before the commencement of the experiment. The blood samples were collected at interval of 2 days (0, 2, 4, 6, 8, 10, 12, 14).
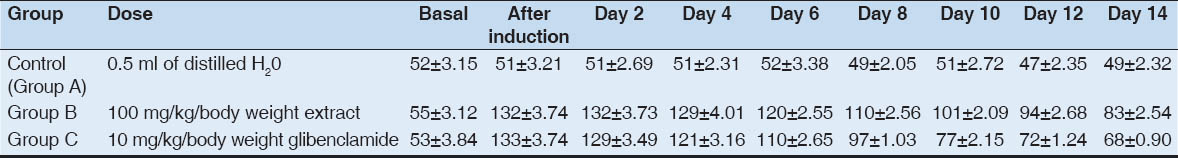
Also hyperglycemia was induced to Group B&C. Group A served as the control group and were administered distilled water. Below is the table for mean blood glucose levels for 10 mg/kg/body weight administration of glibenclamide for 14 days and mean blood glucose levels of extract for 14 days.
Collection of Plant Material
The fresh specimens of V. amygdalina were collected from Mr. Maduka’s Compound, Okofia and were authenticated by a botanist in the Department of Botany, Nnamdi Azikiwe University, Awka, Anambra State.
Preparation of Extracts of V. amygdalina
Large quantities of the fresh specimens of V. amygdalina were washed free of soil and debris. The leaves were air-dried for 3 weeks, and the dried specimens were pulverized using a mechanical grinder. The powder (300 g) was macerated in 1 L of lukewarm water for 48 h. Then, it was filtered with porcelain cloth. It was further filtered using number 1 whatman filter paper. Then, the filtrate was concentrated using oven with extractor fan at a temperature of 50°C. Then, the crude extract was stored in the refrigerator for use.
Procurement of Chemical and Drug
Alloxan and glibenclamide were procured from I.C. Okoye Medical Co. (W.A), D 700 Niger Head Bridge Market, Onitsha, Anambra state.
Induction of Diabetics
Alloxan was used to induce hyperglycemia in male albino Wistar rats. A freshly prepared solution of alloxan 100 mg/kg body weight was injected intraperitoneally. After 24 h of alloxan administration, rats with 43% increase in blood glucose level were considered hyperglycemic and were selected for the experiment.
Experimental Design
A total of 24 adult albino rats were used for the study. They were grouped into three groups of eight animals per group. All the animals were starved 24 h, and their basal fasting blood glucose level were determined using a glucometer. Then, hyperglycemia was induced to Group 2 and 3 by single intraperitoneal administration of 100 mg/kg of alloxan. At 24 h post administration of alloxan, their blood glucose level were checked to confirm hyperglycemia. Then, the animals received treatment thus:
-
Group A: Received 0.5 ml of distilled water as a control group.
-
Group B: Received 10 mg/kg/body weight of glibenclamide.
-
Group C: Received 100 mg/kg/body weight of extract.
At the blood samples were collected at interval of 2 days (0, 2, 4, 6, 8, 10, 12, 14) and administration, blood samples were collected from the animals for the estimation of blood glucose levels.
Determination of Blood Glucose Level
The blood glucose levels of the animals were determined using a glucometer (fine test); blood samples were collected by use of a lancet to puncture the tail vein of the animals. The glucose levels were determined every 2 days of treatment thus 0, 2, 4, 6, 8, 10, 12 and 14 day.
The blood glucose levels were observed at ever 2 days up to 2 wks (0, 2, 4, 6, 8, 10, 12, 14).
Statistical analysis
The data were summarized as Mean ± SD. Groups were compared by repeated measures analysis of variance (ANOVA) and the significance of mean difference within and between the groups was done by Tukey’s post hoc test. A p value less than 0.05 (p<0.05) was considered statistically significant.
RESULTS
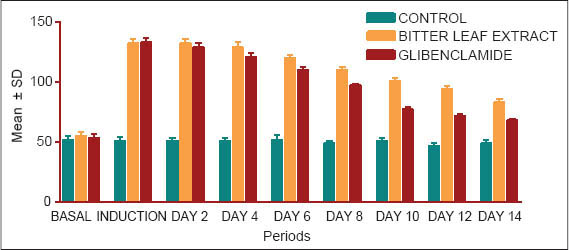
The blood glucose levels of three groups are summarized in Table 1 and also depicted in Fig. 1. Table 1 and Fig. 2 both showed that the mean fasting blood glucose level for extract for 14 days. The extract caused a decrease in blood glucose level on the 4th day, but this was not statistically significant (p>0.05). On the 6th day, the extract caused a slightly significant decrease (p>0.05) in blood glucose level. From the 8 day to the 14th day, a decrease in the blood glucose levels very significant (p>0.01).
Table 1
Effect of glibenclamide and extract on glucose levels was determined every 2 days of treatment
Figure 1
The mean blood glucose levels for extract for 14 days
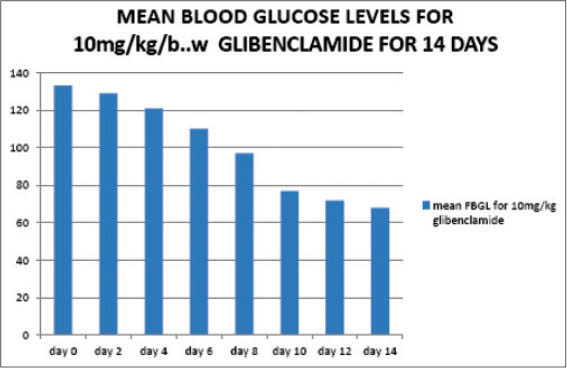
Figure 2
The mean blood glucose levels for 10 mg/kg/body weight of glibenclamide
In contrast, glibenclamide also caused a decrease in the blood glucose level on the third day, however, this decrease was not statistically significant (p>0.05) but slightly significant on the fourth day (p<0.05). On the sixth day, the decrease was moderate significant (p<0.01). From the eight day to the fourteenth day, the decrease in the blood glucose levels became very significant (p<0.001).
Conversely, in control group, it remain similar (p>0.05) over the periods i.e. did not changed significantly.
However, glibenclamide showed significant (p<0.05 or p<0.01) decrease in mean blood glucose level from day 8 to till end (day 14) as compared to bitter leaf extract, suggesting that glibenclamide has a more potent hypoglycemic effect when compared with the extract.
DISCUSSION
Effective control of the blood glucose level is a key step in preventing or reversing diabetic complications and improving the quality-of-life in both Type 1 and Type 2 diabetic patients.[7-9]
The hypoglycemic potentials of medicinal plants have been documented.[6,10] The result of this study confirms the previous report by Akah and Okafor, (1992)[11] on the hypoglycemic effect of the leaf extract of V. amygdalina in rabbits. Although several biologically active constituents were reported present in the extract,[11] it was not demonstrated, which of the groups of phytochemical were responsible for the effect and the mechanism of action. The prompt and remarkable reduction in blood glucose in both the fasting normal rats and alloxan diabetic rats suggest a mechanism of action different from that of sulfonylureas, and unrelated to insulin secretion from pancreatic β-cells. In the present study, V. amygdalina leaf extract present a potent lowering of fasting blood glucose levels in alloxan-induced diabetic rats.
Glibenclamide also known as glyburide is an antidiabetic drug in a class of medications closely related to sulfa drugs. Glibenclamide is a standard drug that is routinely used in the treatment of diabetes.[12] The drug works by binding to and activating the sulfonylurea receptor 1, the regulatory subunit of the ATP-sensitive potassium channels.[13]
The extract caused a decrease in blood glucose level on the 4th day, but this was not statistically significant. Furthermore, glibenclamide caused a decrease in the blood glucose level on the 3rd day; this decrease was not statistically significant but slightly significant on the 4th day.
On the 6th day, the extract caused a slightly significant decrease but when compared with the 6th day of glibenclamide, the decrease was very significant [Table 1].
From the 8 day to the 14th day, a decrease in the blood glucose levels for Group B and C became very significant. However, Group C (glibenclamide) showed highly significant decrease when compared with Group B (extract), suggesting that glibenclamide has a more potent hypoglycemic effect when compared with the extract.
Glibenclamide dose increments above 5-10 mg/day did not significantly add to the blood glucose-lowering effect. Additionally, there was no increased stimulatory effect on insulin secretion at doses beyond 5 mg/day.[14,15] The results of this study are in keeping with evidence suggesting that sufonylureas have maximal blood glucose-lowering effects at doses lower than the manufacturers’ maximum recommended dose. Therefore, using glibenclamide at high doses, eg, 20 mg/day, may not confer increased benefit and may predispose patients to adverse effects, including hypoglycemia and beta cell exhaustion/failure[16].
CONCLUSION
The present study suggests that the glibenclamide has a more potent hypoglycemic effect in alloxan-induced diabetic Wistar rats when compared with the extract and may be more effective in the treatment of diabetics.
REFERENCES
1. Munaya C, Bitter leaf-based extracts cures hepatitis co-inferation and othersThe Guardian Newspaper 2013; July25
2. Iroanya OO, Adebesin OA, Okpuzor J, Evaluation of the hepato and nephron-protective effect of a polyherbal mixture using Wistar albino ratsJ Clin Diagn Res 2014; 8: HC15-21.
3. Schiffers RR, African Indigenous Vegetables: An Overview of the Cultivated Species2013; 3: England: University Greenwich Press; 133-42.
4. Ibrahim G, Abdurahman EM, Ibrahim H, Ibrahim NO, Comparative cytomorphological studies on the studies of V. amygdalina del. and V. kotschyamaNiger J Bot 2010; 23: 133-42.
5. Swee K, Wan Y, Ho B, Boo KH, Woon SL, Huyunh K, An ethnoveterinary used green vegetables with multiple bioactivitiesJ Med Plant Res 2010; 4: 2787-812.
6. Abosi AO, Rasoreta BH, In-vivo anti-malarial activity of V. amygdalina DelBr J Biomed Sci 2003; 60: 89-91.
7. Marble A, Glibenclamide, a new sulphonylurea: Whither oral hypoglycaemic agents?Drugs 1971; 1: 109-15.
8. Abraira C, Colwell JA, Nuttall FQ, Sawin CT, Nagel NJ, Comstock JP, Veterans Affairs Cooperative Study on glycemic control and complications in type II diabetes (VA CSDM). Results of the feasibility trial. Veterans Affairs Cooperative Study in Type II DiabetesDiabetes Care 1995; 18: 1113-23.
9. Ohkubo Y, Kishikawa H, Araki E, Miyata T, Isami S, Motoyoshi S, Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non-insulin-dependent diabetes mellitus: A randomized prospective 6-year studyDiabetes Res Clin Pract 1995; 28: 103-17.
10. Akah P, Obioma N, Ada N, Dorathy A, Effects of aqueous leaf extract of Vernonia amygdalina on blood glucose and triglyceride levels of alloxan-induced diabetic rats (Rattus rattus)Anim Res Int 2004; 1: 90-4.
11. Akah PA, Okafor CL, Blood sugarlowering effects of Vernonia amygdalina del in an experimental rabbit modelPhytother Res 2006; 6: 171-3.
12. Akah PA, Okafor CL, Blood sugarlowering effects of Vernonia amygdalina del in an experimental rabbit modelPhytother Res 2006; 6: 171-3.
13. Katzung BG, Masters SB, Trevor AJ, Basic and Clinical Pharmacology2009; New York: McGraw-Hill Medical;
14. Serrano-Martín X, Payares G, Mendoza-León A, Glibenclamide, a blocker of K+(ATP) channels, shows antileishmanial activity in experimental murine cutaneous leishmaniasisAntimicrob Agents Chemother 2006; 50: 4214-6.
15. Rambiritch V, Naidoo P, Butkow N, Dose-response relationships of sulfonylureas: Will doubling the dose double the response?South Med J 2007; 100: 1132-6.
16. Jonsson A, Hallengren B, Rydberg T, Melander A, Effects and serum levels of glibenclamide and its active metabolites in patients with type 2 diabetesDiabetes Obes Metab 2001; 3: 403-409.